In the next several issues I’ll be explaining the processes incorporated in the modern dry cleaning machine including distillation, washing and extraction, drying, and control. The components used in these processes, and how these components are combined and coordinated to clean garments.
DISTILLATION
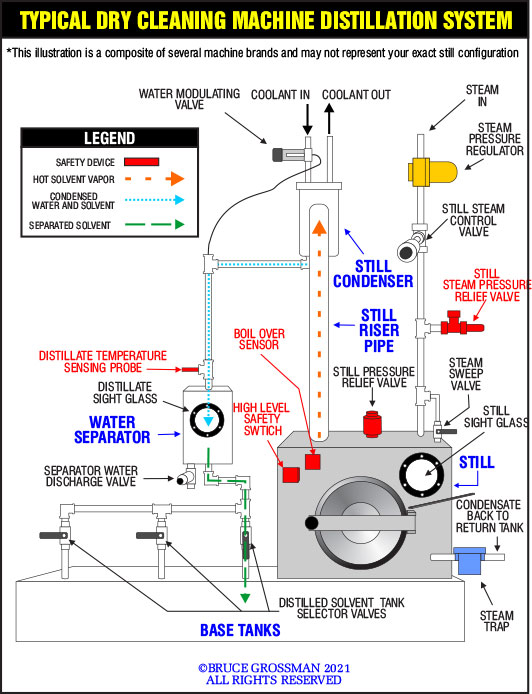 Distillation is a process used to purify a liquid, in our case solvent. Solvent both dissolves impurities it removed from garments like grease, oil, and wax and detergent, (these impurities are called non-volatiles); in addition, solvent suspends solid impurities like lint, dust, and soil, these impurities are called particulates. Proper distillation will in most cases return the distilled solvent to its original condition leaving these impurities behind in the still bottom.
Distillation is a process used to purify a liquid, in our case solvent. Solvent both dissolves impurities it removed from garments like grease, oil, and wax and detergent, (these impurities are called non-volatiles); in addition, solvent suspends solid impurities like lint, dust, and soil, these impurities are called particulates. Proper distillation will in most cases return the distilled solvent to its original condition leaving these impurities behind in the still bottom.
Two steps that comprise the distillation process:
- Evaporating the solvent- Liquid solvent is heated in the still until it boils and vaporizes, changing from a liquid state into a vapor much like steam exiting a teapot.
- Condensing the solvent vapor- Solvent vapor is cooled on some surface (generally a water-cooled coil) much like steam condensing on a bathroom mirror, changing from solvent vapor into a liquid solvent.
EVAPORATING THE SOLVENT
Solvent in the still is vaporized using heat (usually supplied by steam) and in some machines, heat is combined with a vacuum. The addition of a vacuum is used to lower the temperature at which the solvent boils, maintaining the solvent at a temperature below the solvent’s flashpoint (the lowest temperature at which a substance can ignite). For this article, we will use steam as the heating media. Anytime you use steam for heating, a steam trap is required to remove the condensate from the heated vessel, in this case, the still steam jacket (a separate chamber surrounding the still bottom which contains the steam isolating it from the inside of the still which heats the still similar to a flame under a kettle). In addition, there is a steam valve that turns the steam supply on and off. Steam heating also has another very positive attribute, temperature control. Since steam temperature is a function of steam pressure it’s easy to set the heating temperature of the still by setting the steam pressure using a steam pressure regulator.
CONDENSING THE SOLVENT VAPOR
As the solvent is heated and vaporized, the vapor expands and flows up through a large pipe called a still riser and then enters the still condenser. Inside the condenser is a coil of tubing which has cold water flowing through it. When the hot solvent vapor contacts the cold coil it condenses (changes from a vapor to a liquid). Since there is also water present in the solvent when it is added to the still, the water also condenses along with the solvent and must be removed before the solvent can be reused. This is accomplished by the water separator, a device that uses the difference in the specific gravity of the solvent and water to separate the two liquids. The separated solvent flows into a separate tank called the distilled solvent or rinse tank while the water is collected in a separate area to be drained off and disposed of as hazardous waste.
STILL BOTTOMS (NON-VOLATILE RESIDUES AND PARTICULATES)
After the solvent in the still has been boiled away what is left is a combination of waste by-products which must eventually be removed from the still called still bottoms. To minimize the amount of solvent left in these waste by-products, in some stills there is a valve called the steam sweep valve. This valve is used at the very end of the distillation process to conduct steam into the still purging the remaining solvent vapor from the still bottom. In machines using the solvent perchloroethylene, water content contained in the steam forms what’s known as an azeotrope which boils at a much lower temperature than the pure solvent, driving out the last remanent of solvent from the still bottoms.

Bruce Grossman
Bruce Grossman is the Chief of R&D for EZtimers Manufacturing. EZtimers is the manufacturer of the new EZ DOSE boiler compound manager and return tank level control which replaces that troublesome ball float valve in the condensate return tank and automatically adds the correct amount of boiler compound to the return tank preventing the oxygen corrosion and scaling. Our SAHARA and DIB-M high purity separator water mister/evaporators provide a thrifty, legal method to get rid of the separator water generated by your dry-cleaning machine. See our Ad in this issue and for further information on EZtimers products visit www.eztimers.com Please address any questions or comments for Bruce to bruce@eztimers.com or call 702-376-6693.

